226 | Herb fraud shock | Data exchange in traceability |
Plus physical hazard case study
This is The Rotten Apple, an inside view on food fraud and food safety for professionals, policy-makers and purveyors. Subscribe for insights, latest news and emerging trends straight to your inbox each Monday.
Shocking rates of fraud in herbal products
Data exchange in traceability: what you need to know
Food safety news and resources
Case study: Tiny maintenance failure causes huge problems
Food fraud news and incidents
🎧 Listen Now 🎧
Hello, dear reader,
Welcome to Issue 226 of The Rotten Apple, where we discuss herbal preparations: natural, gentle and probably adulterated. In this week’s issue, I unpack a recently published paper about fraud and authenticity methods for herbs. The results are shocking.
Also this week, exactly what you need to know to get ready for the new traceability rules for US businesses, plus a fascinating case study that explores the giant ripples that can be caused when a tiny maintenance failure is overlooked.
Plus, as always, there’s food safety and food fraud news from around the globe, including a new guidance doc from the FAO about foodborne viruses.
Karen
P.S. If you love this newsletter and want to support ad-free, not-boring food reporting, please tell your friends and colleagues about it. Your shares help grow our global food safety community.
Herb Fraud Unpacked
Herbal preparations are often marketed as gentle, natural remedies – but with herbs and spices consistently near the top of the list for food-fraud-vulnerable commodities, what exactly are you really getting when you buy a herbal remedy?
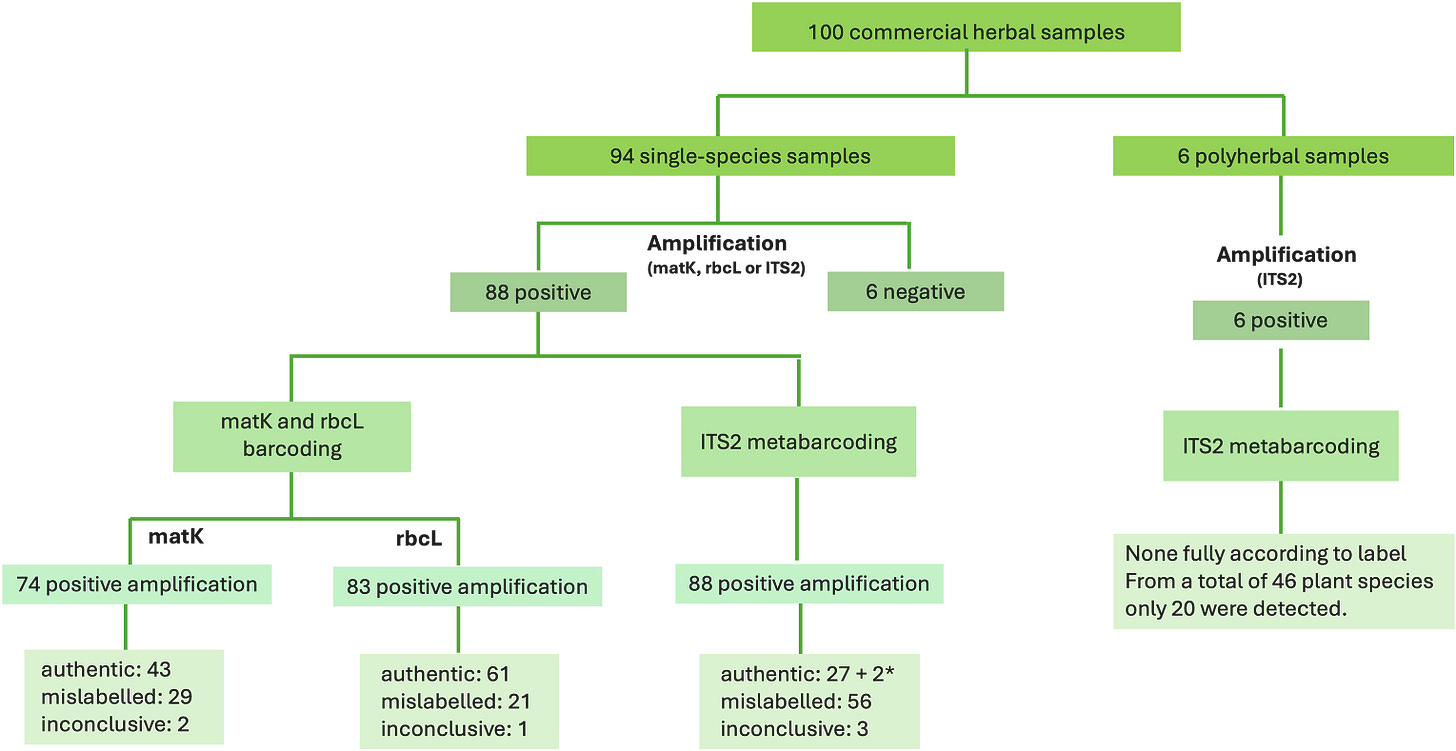
A recent study of herbal products by Portuguese researchers checked the authenticity of 94 products using DNA-based methods, and the results were sobering.
Fraud rates of 66 percent
The team purchased herbal medicinal products labelled as single‑species and multi‑species blends from various online suppliers in Portugal. These products included familiar herbs such as St John’s wort, chamomile, lemon balm, rosemary, fennel, hawthorn, vervain, ginkgo, valerian, angelica, ginseng, guarana, yerba mate, moringa, gotu kola, lemongrass, sweet orange, California poppy and lavender, plus more.
Almost two-thirds of the products were mislabelled (n = 94). Two thirds!
For context, most food fraud surveys of specific commodities find around 10% of samples are fraud-affected. Twenty percent is considered unusually high. Sixty six percent is off the charts.
Among products labelled as single‑species (n = 88), mislabelling affected 64% of samples when judged with the most sensitive approach. That is, 56 of the 88 samples for which DNA amplification was possible were problematic with respect to their composition.
For the multi‑species herbal blends (n = 6), every product (100%) was mislabelled.
In the context of this study, mislabelling means the product was adulterated or diluted with plant material from another species or was entirely replaced by plant material from another species.

Which herbs were the worst?
Among the single‑species products, some herbal preparations were almost never true to label, including:
Ginkgo (Ginkgo biloba) – 100% mislabelled (n = 4)
Muira puama (Ptychopetalum olacoides) – 100% mislabelled (n = 4)
Lemongrass (Cymbopogon citratus) – 100% mislabelled (n = 3)
Ginseng (Panax ginseng) – 100% mislabelled (n = 1)
St John’s wort (Hypericum perforatum) – 83% mislabelled (n = 6)
Other products were affected by mislabelling less frequently but still at unacceptably high rates, including:
Valerian (Valeriana officinalis) – 50% mislabelled (n = 4)
Siberian ginseng (Eleutherococcus senticosus) – 50% mislabelled (n = 4)
Which herbs were okay?
A small group of botanicals appeared free of mislabelling in this study, with all samples of the following being true to label:
Chamomile (Matricaria chamomilla) – 0% mislabelling (n = 3)
Lemon balm (Melissa officinalis) – 0% mislabelling (n = 3)
Rosemary (Rosmarinus officinalis) – 0% mislabelling (n = 2)
Angelica (Angelica archangelica) – 0% mislabelling (n = 1)
Hawthorn (Crataegus monogyna) – 0% mislabelling (n = 2)
So, using this single study as a guide, if you purchased chamomile or lemon balm, you were much more likely to get what the label promises than if you bought, say, ginkgo, St John’s wort or ginseng.
Testing complexities
I’ve just given you the simple version of the results. But as with all things food science, the realities are more complex. The results were different depending on which method the researchers used. In fact, the main aim of their research was to compare three different DNA-based tests for authenticating herbal infusions.
The three methods they used were matK DNA barcoding, rbcL DNA barcoding and ITS2 metabarcoding. Additionally, for samples analysed with ITS2, the researchers also compared two reference databases: NCBI and BOLD.
Firstly, not every sample had DNA that could be ‘seen’ in the tests. The tests used in this study rely on any DNA in the samples being amplified using the polymerase chain reaction (PCR), as an important part of the testing process. The amplification step produces many copies of specific DNA regions so that they can be sequenced and compared against reference databases to identify the plant species present.
There were 6 products among the 100 the researchers purchased that had to be excluded from the results because DNA amplification did not work. This could be because those products contained no ‘readable’ sections of DNA, as would be the case if the herbs had been very intensively processed, or if they contained significant quantities of PCR inhibitors, which can include polyphenols, polysaccharides and caffeine‑like compounds, or if the products were not derived from plant material.
Among the samples that could be sequenced by at least one method, not all of them could be sequenced using the other methods and/or databases.
Shockingly, around half of the samples that would have been considered authentic if DNA barcoding alone was used for authentication also contained additional undeclared species.
So for single‑species-labelled products, the probable fraud (mislabelling) rates varied significantly according to the method used, as follows:
matK barcoding: 39%
rbcL barcoding: 25%
ITS2 metabarcoding: 64%
The researchers concluded that conventional DNA barcoding (matK and rbcL) tended to overestimate authenticity, because it often failed to detect undeclared species present in the products.
ITS2 metabarcoding, by contrast, was able to pick up multiple species in the same product and gave approximate quantities, allowing the researchers to see which species were present as major or minor components of each product. In this set of tests, one of the ITS2 reference databases clearly outperformed the other, providing more complete and reliable identifications.
A final complexity was that some samples appeared to contain plants from an unlikely genus, which turned out to be a misidentification, with the sequences ultimately found to be from fungi present in the products.
Safety concerns
The ITS2 metabarcoding method revealed fungal DNA in many of the samples. Fungi are naturally present as spores or mycelium on harvested plants, and fungal contamination is a known problem in botanicals. The genuses present in this set of samples included fungi known to produce mycotoxins, such as Aspergillus and Penicillium.
Earlier work showed that nearly 84% of samples of herbal teas and medicinal plants contained at least one mycotoxin, with the highest levels of aflatoxins and zearalenone detected in chamomile flowers, the second most consumed medicinal plant in Portugal.
Across all the samples in the earlier study, organic samples and materials sold in bulk had the highest levels of zearalenone, a mycotoxin produced by Fusarium species.
Takeaways for food professionals
The results of this single study are pretty shocking: the label was wrong for almost two-thirds of the medicinal herbs and herb mixtures the researchers purchased.
It’s likely a global phenomenon: While I’ve only discussed the results from this study, which tested samples purchased by researchers in Portugal, it’s likely that similar results would be seen in products purchased elsewhere, because medicinal botanicals have global supply chains.…. St John’s wort purchased in Portugal could plausibly have the same origin as St John’s wort purchased in Cincinnati, with production and extraction occurring in places such as China, Chile, and various European countries, then exported worldwide as the bulk herb or extract.
Method matters: simple DNA barcoding can under‑detect adulteration, while ITS2 metabarcoding revealed hidden complexity and undeclared additional components.
Not all herbs are equal: some species are more vulnerable to substitution and adulteration than others. In this set of samples ginkgo, muira puama, lemongrass, St John’s wort, ginseng and valerian were more problematic than chamomile, lemon balm, rosemary, angelica and hawthorn.
‘Natural’ does not mean pure or safe: the frequent detection of non‑labelled plant species and fungi, including mycotoxin producers, shows why robust supply chain controls and authenticity testing are essential for botanical products.
In short: A recently published study that explored the authenticity of herbal medicinal products revealed a shocking rate of mislabelling with respect to species and purity 🍏 A sub-set of well-known herbs exhibited mislabelling in 100% of products purchased for this study, while a few were found to be consistently true to label 🍏 The researchers reported significant differences between results gained with DNA barcoding and metabarcoding methods and between databases used with the metabarcoding method 🍏 Fungal DNA was recovered from many samples, revealing potential mycotoxin hazards 🍏 The results are broadly extrapolatable to herbal products globally due to the nature of supply chains for such products🍏
Main source:
Pinto, M.A., Santos, J., Quaresma, A., Honrado, M., Rufinao, J., Amaral, J. (2026) ‘Comparison of DNA barcoding and metabarcoding approaches for the authentication of herbal infusions in the Portuguese market’, Food Control, 183, 111893. Available at: https://www.sciencedirect.com/science/article/pii/S0956713525007625
What you need to know about data exchange for FSMA traceability
A guest post by John Henry Wells, PCQI lead instructor and food traceability expert
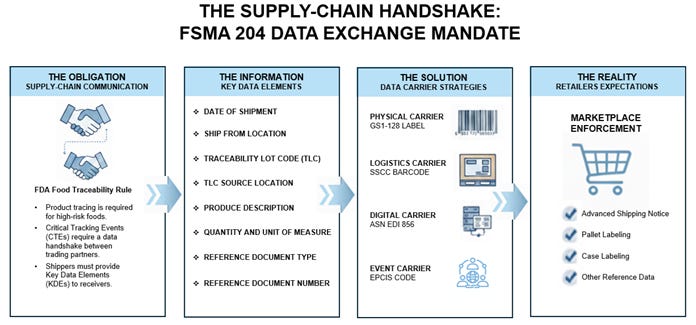
The Supply-Chain Handshake
The Obligation: It’s About Supply-Chain Communication
Section 204 of the Food Safety Modernization Act (FSMA), the Food Traceability Rule, directed the United States Food and Drug Administration (FDA) to move the United States from a reactive to a proactive food safety posture through enhanced product traceability.
The mandate authorised the FDA to establish a system to “effectively and rapidly track and trace food” both domestically and for imports, with the aim of preventing the spread of foodborne illness.
The FDA Food Traceability Rule requires a product tracing system for high-risk foods, and imposes strict recordkeeping rules to ensure that when an outbreak occurs, the agency can rapidly identify and investigate suspect food to prevent illness and death.
To achieve its product tracing aim, the FDA considered supply-chain Critical Tracking Events (CTEs) no longer a siloed activity, but as points that require a handshake between trading partners. While the FDA allows flexibility in how records are kept, they are rigid about the fact that specific Key Data Elements (KDEs) must be passed forward at the time of a CTE.
The FDA Food Traceability Rule treats Critical Tracking Events (CTEs) not as isolated activities, but as distinct points of contact that require a handshake between trading partners.
For every CTE, the Food Traceability Rule requires the purveyor to maintain records and provide specific information to the immediate subsequent recipient. When you ship a food listed on the Food Traceability List (FTL), you are not just moving physical inventory; you are legally required to move data to ensure there is an unbroken traceability thread through the supply chain.
The Information: The Shipper’s Message to the Receiver
According to 21 CFR 1.1340(b), the burden of continuity falls on the shipper. You must provide the immediate subsequent recipient (the receiver) with specific Key Data Elements (KDEs). Without this transfer, the receiver cannot fulfil their own compliance obligations. The KDEs include:
Date of Shipment
Location Description for the “Ship From” Location
Traceability Lot Code (TLC) - This is the single most critical piece of data. You must send the exact code (often alphanumeric) assigned to the specific lot of food being shipped. You generally cannot change this code during a Shipping CTE. You simply pass along the code you received, unless you are the one who initially created the lot.
Location Description for the Traceability Lot Code (TLC) Source - This is the most frequently missed KDE. If you are not the one who assigned the Traceability Lot Code (i.e., you are a distributor, cross-docker, or warehouse), you must tell the receiver who originally assigned the lot code. You must pass this information forward from the person who sent it to you. If you assigned the lot code (e.g., you are the manufacturer or packer), then you are the TLC Source.
Product Description
Quantity and Unit of Measure
Reference Document Type and Number (e.g., Bill of Lading #12345)
Together, these Key Data Elements for each Critical Tracking Event create supply-chain dependency. If you are a manufacturer, your customers’ compliance is contingent on your ability to transmit this data. The FDA emphasises that shippers must provide this information in a way the receiver can use. That is, there must be a supply-chain virtual handshake to make this work.
The Solution: Data Carrier Strategies
While the FDA rule is technically technology-neutral, the industry has largely decided that neutrality does not scale. To avoid chaos, major trade associations, including the Produce Traceability Initiative (PTI), the Food Industry Association (FMI), and the Global Dialogue on Seafood Traceability (GDST), have aligned around the GS1 System of Standards.
The predominant data carriers identified to facilitate the exchange of KDEs include:
Physical Carrier - For case-level traceability, the GS1-128 barcode is the workhorse. PTI guidance specifically recommends this format to encode the GTIN (Global Trade Item Number) and Batch/Lot Number. This strategy bridges the physical and digital worlds and allows a barcode scanner to capture the KDEs, reducing manual entry errors.
Logistics Carrier - For pallets and logistics units, the Serial Shipping Container Code (SSCC) is the critical license plate. FMI and GS1 advise using an SSCC on a pallet label to link the physical shipment to the electronic data. One scan of an SSCC on a receiving dock can unlock all the lot-level detail contained in an Advance Ship Notice (ASN), eliminating the need to scan every individual case.
Digital Carrier - An Advance Ship Notice (communicated via EDI 856 data standards) has emerged as a utilitarian method for transmitting transactional data between trading partners. This is an efficient option to capture and share item details and Batch/Lot numbers. Data segments within the ASN allow the transmission of required FSMA 204 KDEs, including the Traceability Lot Code and TLC Source, before the truck even hits the dock.
Event Carrier - For more advanced visibility, Electronic Product Code Information Services (EPCIS) is the standard for sharing the ‘what, where, when, and why’ of an event. GDST leverages EPCIS attributes to bridge gaps between sector standards and FSMA 204 requirements.
The Reality: What Retailers Expect Now
While the FDA has signalled enforcement discretion until 2028, commercial enforcement is happening now.
Major US retailers have already communicated specific traceability requirements to suppliers to ensure interoperability across their distribution networks. If you are waiting for the Federal deadline to upgrade your handshake systems, you are already late by market standards.
Their expectations include:
Electronic Transmission: Mandatory EDI 856 ASN for every shipment.
Pallet Labelling: Pallets must carry an SSCC barcode linked to the ASN.
Case Labelling: Cases must include a GS1-128 barcode with the GTIN and Lot Number.
Marketplace Deadline: Many of these retailers have set compliance dates as early as January 1, 2027, specifically to troubleshoot technical integration before the FDA’s final enforcement begins.
While the FDA permits paper records, the market does not. Retailers are pushing for electronic data to automate receiving at distribution centres. If your traceability plan relies on emailing PDFs, you may be compliant with the law, but you will be out of sync with your biggest customers.
Where Do You Stand?
Commentators report a bifurcation across the industry: those treating traceability as a compliance checkbox, and those treating it as a digital infrastructure project.
Are you ready for the new age of digital traceability, or are you and your suppliers falling behind?
About the author: John Henry Wells, D. Engr., PCQI, is a leading authority in food safety and perishable food handling. With over 40 years of food industry experience spanning research, manufacturing, distribution, and retail, he integrates technical rigour with practical operational insight.
Contact John Henry Wells through LinkedIn and consider following his weekly newsletter.
This article first appeared on LinkedIn.
This week’s food safety news and resources (which are hand-curated, always free and never boring) has updates on the cereulide infant formula situation, mysterious reports of arsenic in candy and good news for antimicrobial resistance in Australian meat (plus more).
Click the preview box to read….
Food Safety News and Resources | February
16 February |
☠ Cereulide baby illnesses reported, plus expanded recall (Global)
🤒 Outbreaks: Salmonella in pistachio and pecans (Canada)
🍬Arsenic in candy (USA)
⚠ Recall: enoki mushrooms for Listeria (Canada)
🦠 Antimicrobial-resistant bacteria in raw meat: good news (Australia)
📖 New guidance: Foodborne viruses (FAO)
🎓Webinar - Analysis and Prevention of Foreign Body Recalls, 18th February
Case Study: Tiny maintenance problem causes withdrawals across 16 countries
In early November 2024, a food company in Germany discovered tiny strands of copper in one batch of its product: 10 kg buckets of mayonnaise. Some of the batch had been distributed to customers in six EU countries, so the company commenced a recall.
By mid-November 2024, a second safety alert had been posted for food(s) packed in buckets, produced by a different company. These were also contaminated with fragments of copper. The copper fragments were tiny, less than 0.16 mm in diameter and 0.1-20 mm long. Fragments of this size are too small to be detected by metal detectors.
Because the same issue affected two unrelated manufacturers, investigators suspected that the buckets themselves were the source of the contamination. They visited the bucket-making site and found that an earthing cable in the energy chain associated with the automatic bucket-stacking machine had become brittle and frayed from constant bending.
Cracks in the plastic sheath of the cable had allowed small strands of copper to escape and they had found their way into the buckets during stacking.
Once this was confirmed, it was revealed that a second type of bucket could have been affected in the same way. Because the buckets were used for holding ingredients like mayonnaise, cream cheese, yoghurt, and sour cream that are used in many foods, multiple withdrawals of final products had to occur in 16 EU countries.
This incident shows how the equipment in suppliers’ facilities can create unexpected physical contamination hazards for food businesses. Even small failures, like a few cracks in the sheath of a simple electrical cable, can cause large problems for food manufacturers and their customers.
In this case, the source of the contamination - the damaged cable - was at a bucket-making facility, nowhere near food, and so less likely to be subject to the close inspection and strict hygienic design criteria required for food equipment.
A further failure was the food manufacturers’ use of the buckets without first cleaning or inspecting them for the presence of foreign material. It is common practice not to clean or check such containers if the supplier holds an appropriate food safety certification, as these certifications are designed to give assurance that the supplier operates systems to prevent contamination.
Together, these three failures – the failure to discover the shedding of copper fragments at the machine, the failure of the bucket supplier to identify the contamination before shipping the buckets, and the failure of the food manufacturers to discover the contamination before placing food in the buckets – led to large and unexpected consequences that touched hundreds of food businesses.
Sources:
European Commission (2025) Health and Food Safety 2024 Annual Report Alert & Cooperation Network. doi: https://doi.org/10.2875/0397221
European Commission (2024) Foreign bodies (copper strands) in salad mayonnaise from Germany https://webgate.ec.europa.eu/rasff-window/screen/notification/724691
European Commission (2024) Foreign bodies (pieces of copper strands) in packaging buckets from Germany https://webgate.ec.europa.eu/rasff-window/screen/notification/725673
🍏 More like this: Frogs and Other Foreign Objects in Food; Physical Hazards Explored | Issue 118 🍏
Below for paying subscribers: Food fraud news, horizon scanning and incident reports
📌 Food Fraud News 📌
In this week’s food fraud news:
📌 Methods for dairy food speciation and herbal preparations
📌 Warnings for supply chains in certain locations
📌 Fraud in buffalo ricotta
📌 Bad results for goat cheese